Introduction
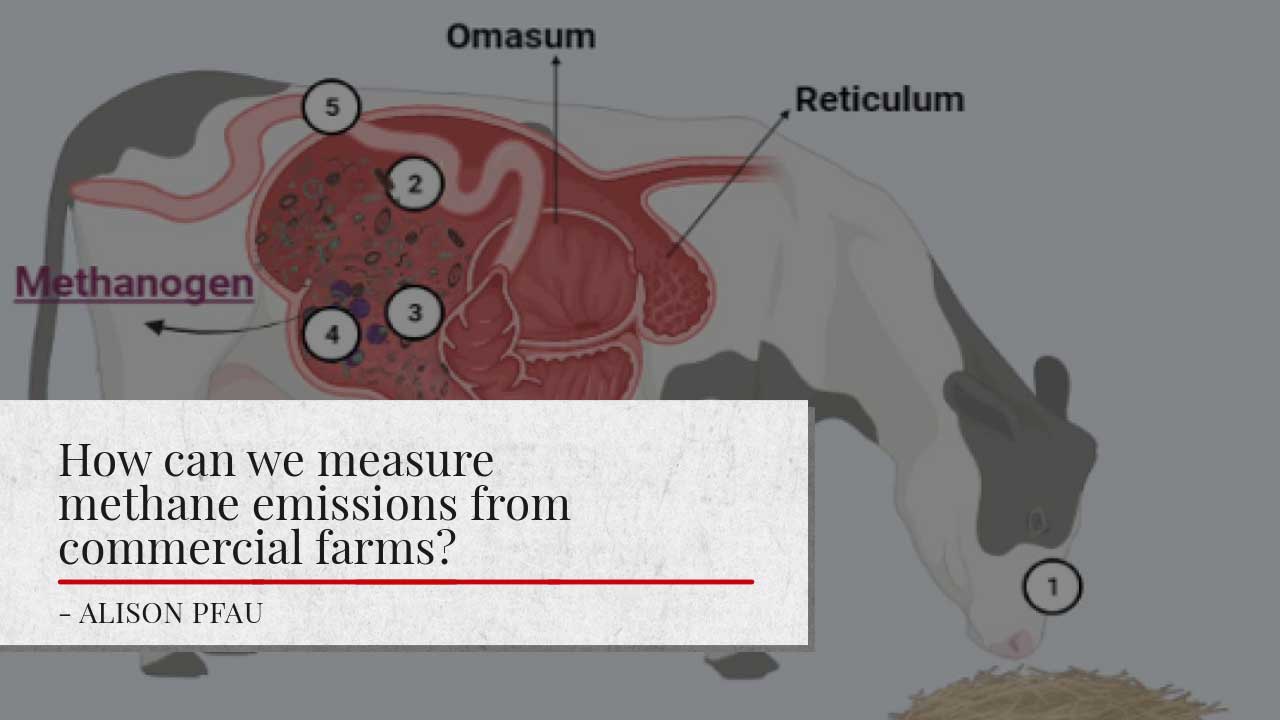
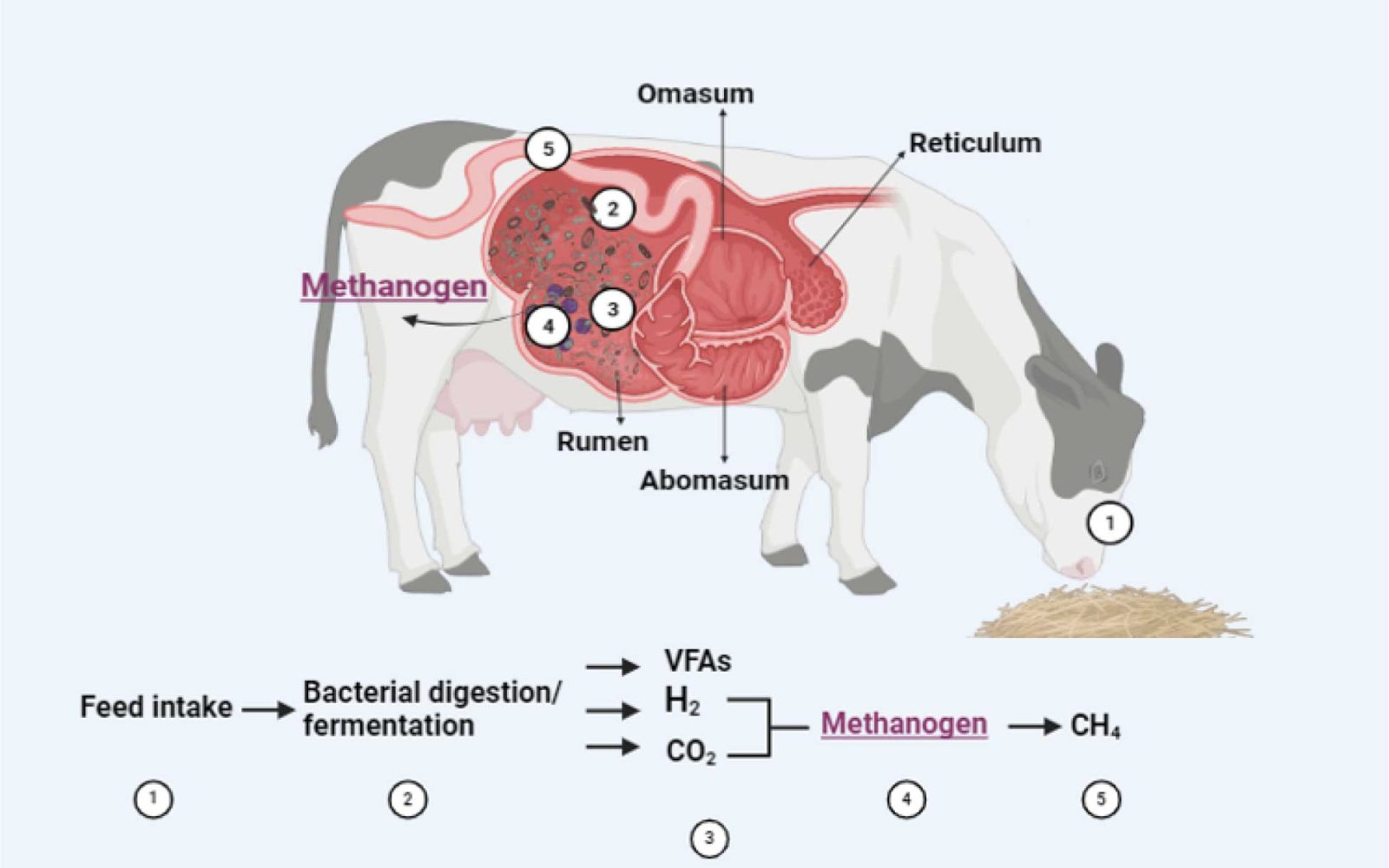
Based on their symbiotic relationship with the rumen microorganisms, dairy cows have an extraordinary capacity to transform human-inedible plant materials into energy for maintenance and lactation. However, the sub-product of this rumen fermentation is methane emissions. Overall, we have two main sources of methane emissions in a farm, one is enteric fermentation that predominantly produced in the digestive tract of ruminant animals, and manure management that it is the methane produced by methanogenesis during manure storage (Garnsworthy et al., 2019)
Methane from the enteric fermentation represents around 30-40% of the gasses in the rumen, which are expelled from the animal by belching. The importance of this gas production is increasing due to the large number of animals and the type and amount of feed they are consuming, producing most methane gasses per unit of feed consumed (Figure 1).
Studies around the world confirm that the lifespan of methane compared to the other greenhouse gases is short, meaning that methane is degraded in the atmosphere after about 10 years, a blink compared with carbon dioxide, which can linger for thousand of years. However, methane still is a potent cause of global warming, and the dairy farmers and academics are constantly working together to find different alternatives to mitigate methane from farms while keep the production and the animal welfare.
To find these alternative ways to reduce enteric methane emissions by ruminants, researchers and farmers have used several methods to measuring methane output by individual dairy cattle under various environmental conditions. These methods included respiration chambers, the Sulphur hexafluoride (FS6) tracer technique, Greenfeed system, Laser methane detector and Sniffers (Table 1).
Table 1. Summary of the main features of methods for measuring methane output by individual1 animal.
(Adapted from Garnsworthy et al., 2019)
| Method | Purchase cost2 | Running Cost2 | Labor/Maintenance2 | Repeatability | Behavior alteration3 | Animal Throughput | Gasses Measured |
| Respiration chamber | High | High | High | High | High | Low | CH4, CO2, H2, O2 |
| SF6 Technique | Medium | High | High | Medium | Medium | Medium | CH4 |
| Sniffer – breath sampling during milking and feeding | Low4 | Low | Low | Medium | None | High | CH4, CO2, H25, O2 |
| Greenfeed | Medium | Medium | Low | Medium | Low | Medium | CH4, CO2, H2, O2 |
| Laser methane detector | Low | Low | High | Low | Low-Medium | Medium | CH4 |
Methods for measuring enteric methane
Respiration chamber
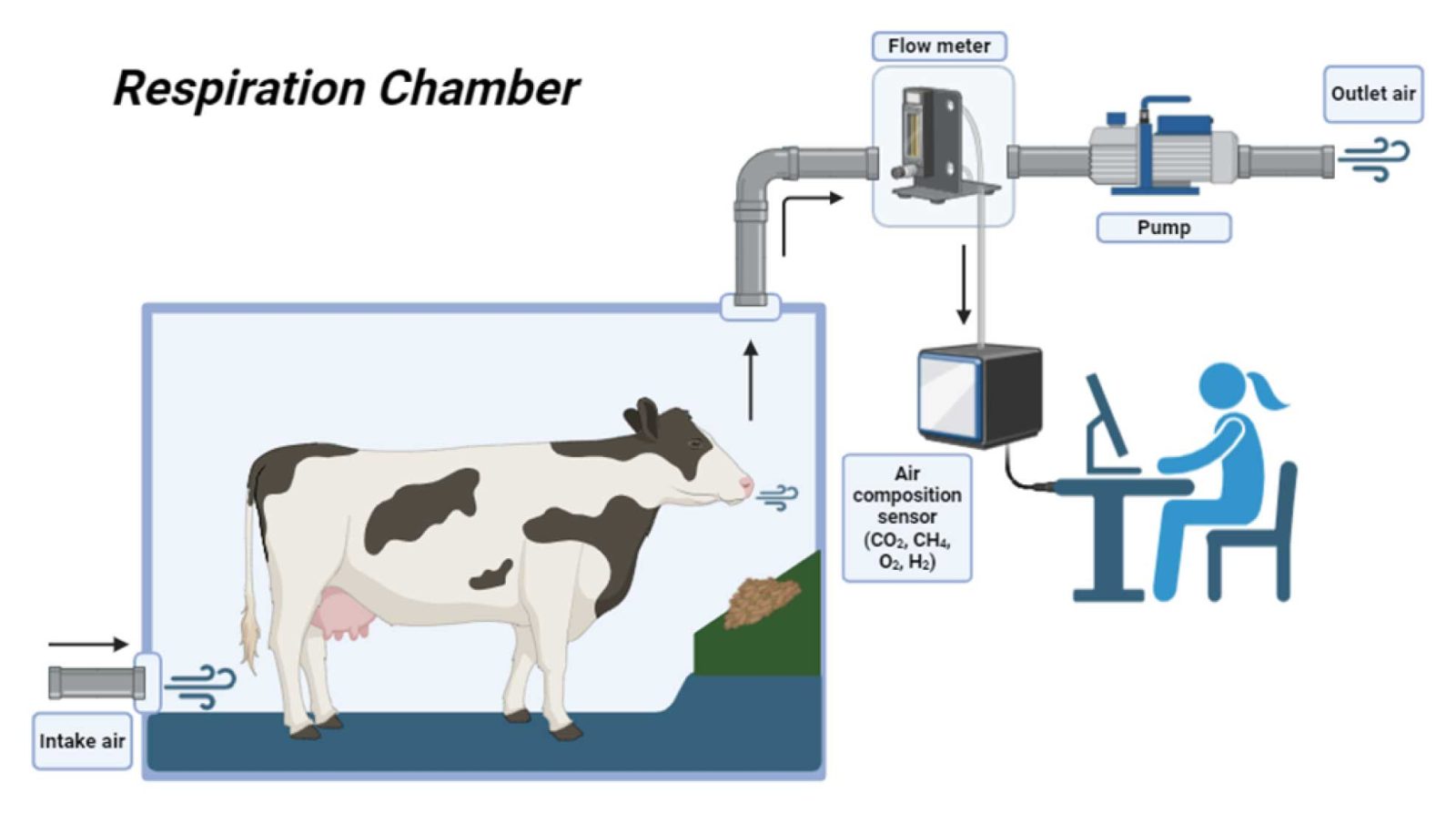
The primary objective of this method is to measure the incoming and exhaust airflows, the concentration of target gases in the air, and the initial and final gas concentrations within the chamber. The animal needs to acclimate to the chamber environment before any measurements are taken. Following this acclimatization period, the animal should remain in the chamber for at least 3 days, during which time it is fed and milked, and any manure must be removed from the chamber.
Benefits
- The method provides accurate results when properly calibrated and operated.
- The method captures all methane emissions, including those from the anus.
- Measurements are taken continuously over several days, accounting for diurnal variations in methane emissions.
Limitations to consider
- Animals are placed in an “unnatural” environment, often resulting in stress and decreased feed intake and/or milk production.
- Chambers cannot accommodate a large number of large animals simultaneously, limiting their usefulness for genetic selection of low-methane emitters.
- They are expensive and require extensive training and specialized operational skills.
Sulfur hexafluoride (SF6) tracer technique
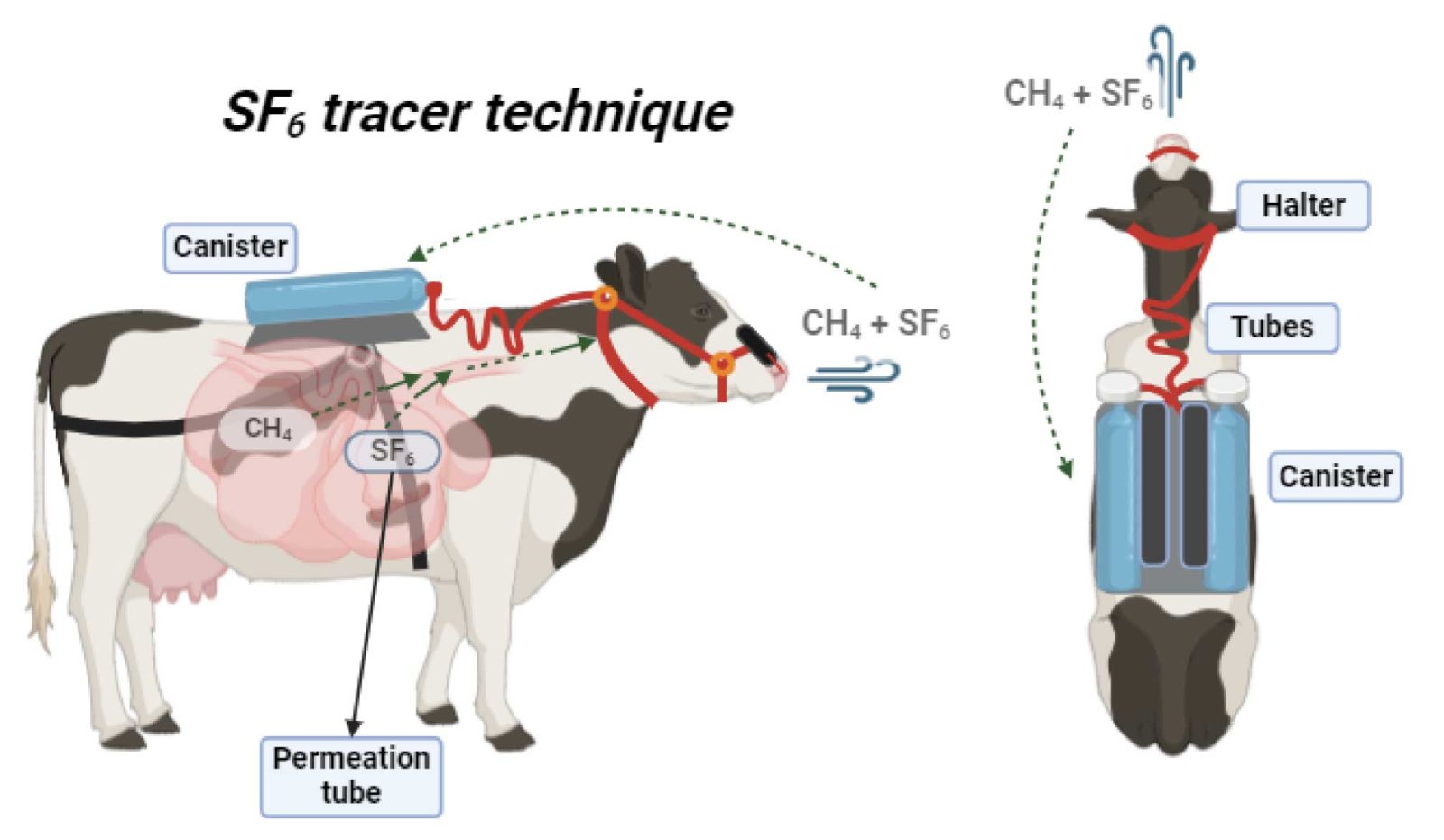
This method involves using a tracer gas, specifically SF6, which is continuously released at a known rate from a permeation tube placed in the reticulum (one of the compartments of the ruminant stomach) and mixed with rumen gases. As the animal eructates and exhales, the gas is continuously sampled around its nostrils into an evacuated container, and the gas is analyzed for SF6 and methane content. Animals need to be measured for at least 5 to 7 days, and it is recommended that group sizes be limited to fewer than 15 animals.
The ratio of the two gases, along with the known SF6 release rate, is used to calculate the methane emission rate. This technique is less costly compared to the use of respiration chambers.
Benefits
- The SF6 technique can be used in a large number of animals in their natural environment
- The SF6 technique can produce accurate methane emission data from a large group of animals
Limitations to consider
- The technique does not account for methane production from the hindgut and excreted through the rectum.
- Variability with the technique has historically high, but the modification created by Deighton have addressed the most important sources of error, generating more accurate data.
- Animal behavior and intake might be compromised by wearing the equipment and by daily handling to exchange canisters.
When using the SF6 technique, is necessarily follow some important conditions to reduce variability in the methane measurements:
- Ensure a high release rate of SF6 from the permeation tube
- Conduct measurements over at least five consecutive days
- Maintain low concentrations of SF6 and methane in the background air, meaning the technique should not be used in enclosed barns unless there is adequate ventilation throughout the measurement period.
GreenFeed
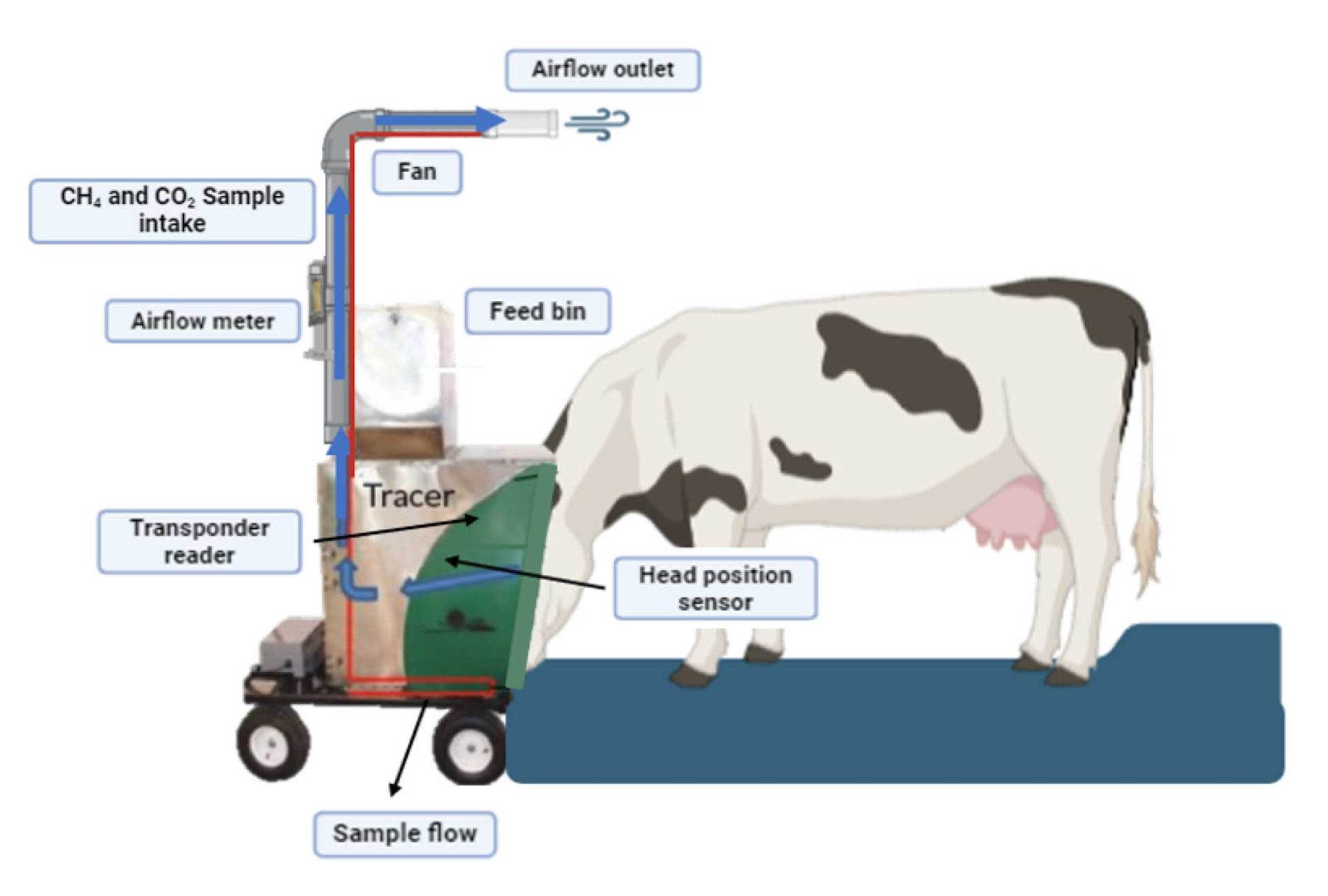
Green-Feed (GF) is a head chamber system developed for multiple spot sampling of exhaled and eructated gases. The method attracts animals to the GF, where they remain for 3-5 minutes on average during each sampling event while the eructated and exhaled gases are captured and directed to gas sensors.
It is recommended that the GF be available 24-7, because methane emissions exhibit a clear diurnal pattern related to feed intake. The system provides the methane emissions from each cow in grams per day.
Benefits
- Portable, user-user and animal-friendly
- It measures methane emissions in grams per day (g/day), compared to another method that only measures methane concentration (e.g., Sniffers).
- GF is an efficient tool to collect data from a large number of animals in a short period.
Limitations to consider
- The technique only measures methane emissions when the animals have their heads in the feeder and are eating, so correlations with whole-day emissions must be thoroughly examined
- Some animals may not be interested in using the system
- Multiple GF units are required for highly stocked pens or pasture
Sniffer
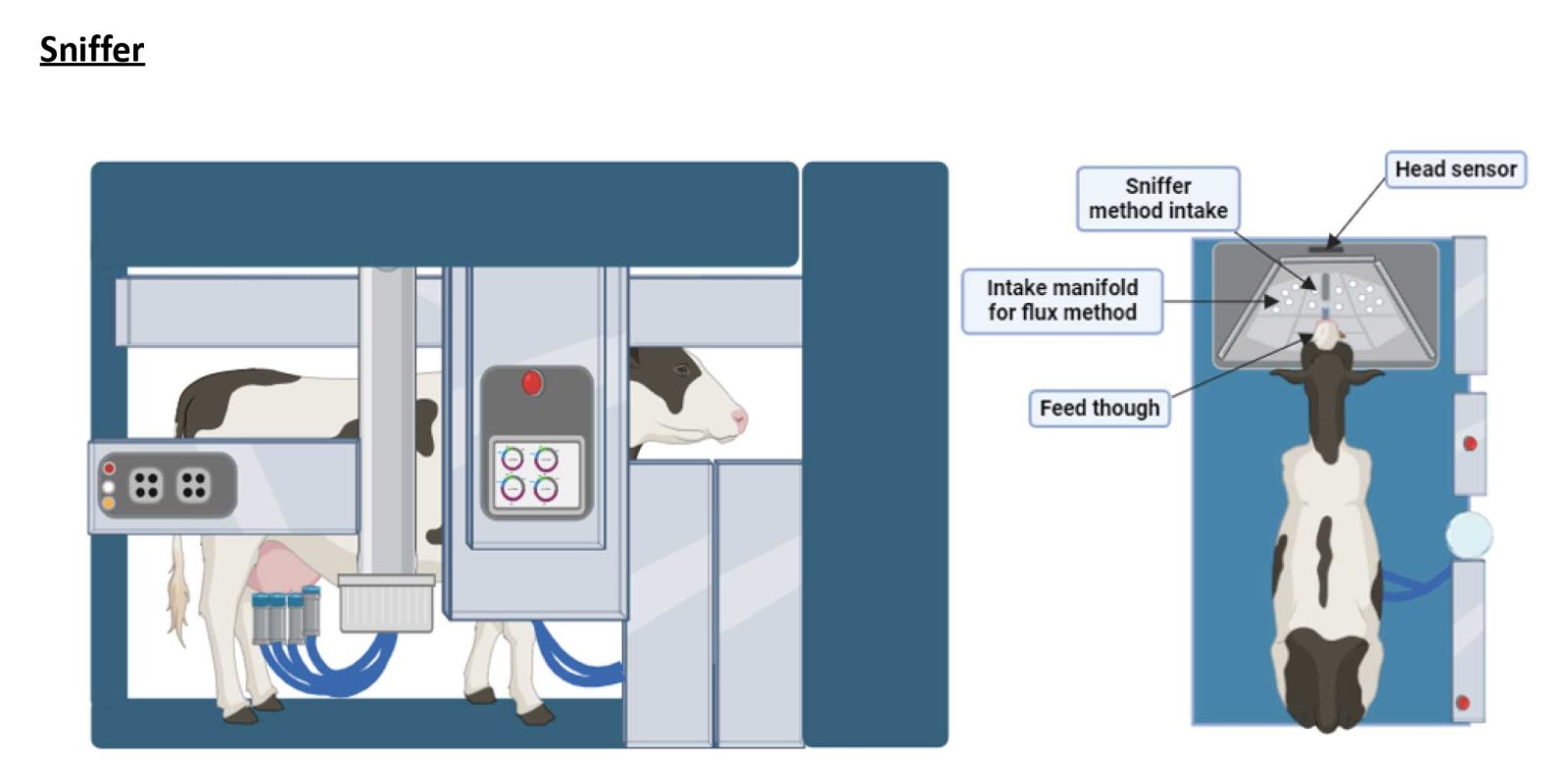
Sniffer is an indirect method that works by estimating methane emissions during an eructation event and the frequency of those eructation events during the measurement period. This method involves collecting air samples directly from the feed manager of an automated milking system during milking. This method utilizes a tube positioned near the cow’s nostrils within the feed bin. This tube connects directly to a gas analyzer, which measures methane concentration during milking sessions typically lasting 3 – 10 minutes.
The overall mean of this method is to calculate the average methane concentration throughout the sampling period. Some research centers measure carbon dioxide (CO2) alongside methane as an additional technique to estimate daily methane output by comparing the methane to CO2 ratio with a predicted CO2 output based on the cow’s performance.
Benefits
- Measurements can be conducted directly on the farm, eliminating the need for specialized facilities
- The method allows for testing a larger number of animals compared to individual animal monitoring
- The equipment does not disrupt the animals’ routine or environment, minimizing stress and allows for natural behavior during milking and feeding
- Reduced handling and training, minimizing disruptions and simplifies the process
- The method is generally affordable, with running costs being relatively low.
Limitations to consider
- The method does not capture methane exhaled directly from the lungs
- Factors like fluctuating air flow in the barn and head position of the animal can impact the accuracy of measurements across different animals
- Raises concerns about the sniffer method’s ability to accurately measure and rank methane emissions among individual cows.
- The number of measurements per analyzer depends on the number of cows assigned to a specific milking station or feeding area and the duration of equipment installation.
Conclusion
Dairy farmers and researchers are actively collaborating to develop effective strategies for reducing methane emissions from dairy cattle. While numerous methods exist to accurately measure and estimate methane output from individual cows and general cattle populations, challenges arise when scaling these methods to large herds. High costs associated with some measurement techniques hinder their widespread adoption.
However, promising technologies like SF6 and Green-Feed and sniffers offer potential solutions for large-scale, cost-effective methane assessment, particularly in extensive grazing systems. The industry anticipates increased accessibility of these methods in the coming years, facilitating broader methane measurement and mitigation efforts on dairy farms.
References
Garnsworthy PC, Difford GF, Bell MJ, Bayat AR, Huhtanen P, Kuhla B, Lassen J, Peiren N, Pszczola M, Sorg D, Visker MHPW, Yan T. Comparison of Methods to Measure Methane for Use in Genetic Evaluation of Dairy Cattle. Animals (Basel). 2019 Oct 21;9(10):837. doi: 10.3390/ani9100837. PMID: 31640130; PMCID: PMC682646
Garnsworthy PC, Craigon J, Hernandez-Medrano JH, Saunders N. On-farm methane measurements during milking correlate with total methane production by individual dairy cows. Journal of Dairy Science. 2012;95(6):3166–3180
Zhao, Yiguang, et al. “A review of enteric methane emission measurement techniques in ruminants.” Animals 10.6 (2020): 1004. https://doi.org/10.3390/ani10061004
https://www.ncbi.nlm.nih.gov/books/NBK519293
Storm IM, Hellwing AL, Nielsen NI, Madsen J. Methods for Measuring and Estimating Methane Emission from Ruminants. Animals (Basel). 2012 Apr 13;2(2):160-83. doi: 10.3390/ani2020160. PMID: 26486915; PMCID: PMC4494326.
Coppa, M., J. Jurquet, M. Eugène, T. Dechaux, Y. Rochette, J. M. Lamy, A. Ferlay, and C. Martin. 2021. Repeatability and ranking of long-term enteric methane emissions measurement on dairy cows across diets and time using GreenFeed system in farm-conditions. Methods. (186):59-67. doi: 10.1016/j.ymeth.2020.11.004
Chagunda, M. G. G., D. Ross, and D. J. Roberts. “On the use of a laser methane detector in dairy cows.” Computers and Electronics in Agriculture 68.2 (2009): 157-160.
Fresco, S., D. Boichard, S. Fritz R. Lefebvre, S. Barbey, M. Gaborit, P. and Martin P. 2023. Comparison of methane production, intensity, and yield throughout lactation in Holstein cows. J Dairy Sci. 106(6):4147- 4157. doi: 10.3168/jds.2022-22855.
Wu L, Koerkamp PWGG, Ogink N. Uncertainty assessment of the breath methane concentration method to determine methane production of dairy cows. Journal of Dairy Science. 2017;101(2):1554–1564







